Priority Disease Areas
With a focus on the Psychiatry & Neurology and Oncology as priority disease areas, Sumitomo Pharma Group will contribute to the betterment of healthcare and fuller lives of people worldwide through diverse approaches, including pharmaceutical products, regenerative medicine/cell therapy, and non-pharmaceutical solutions.
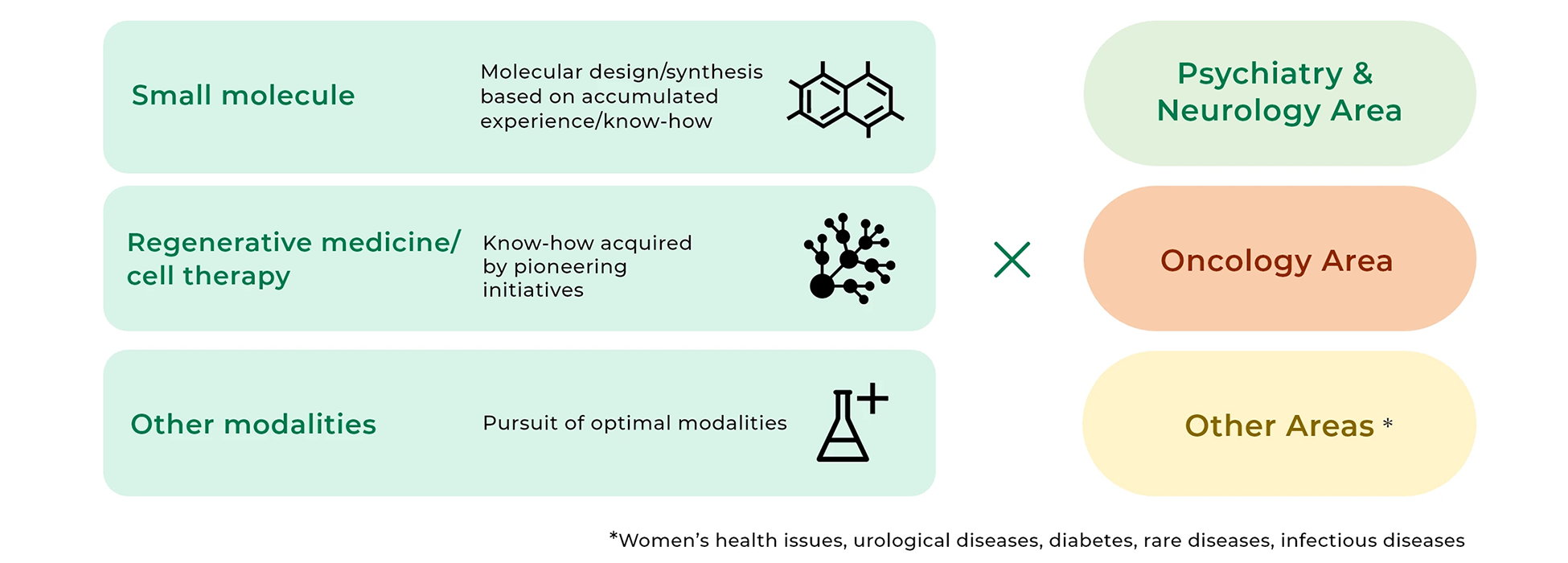
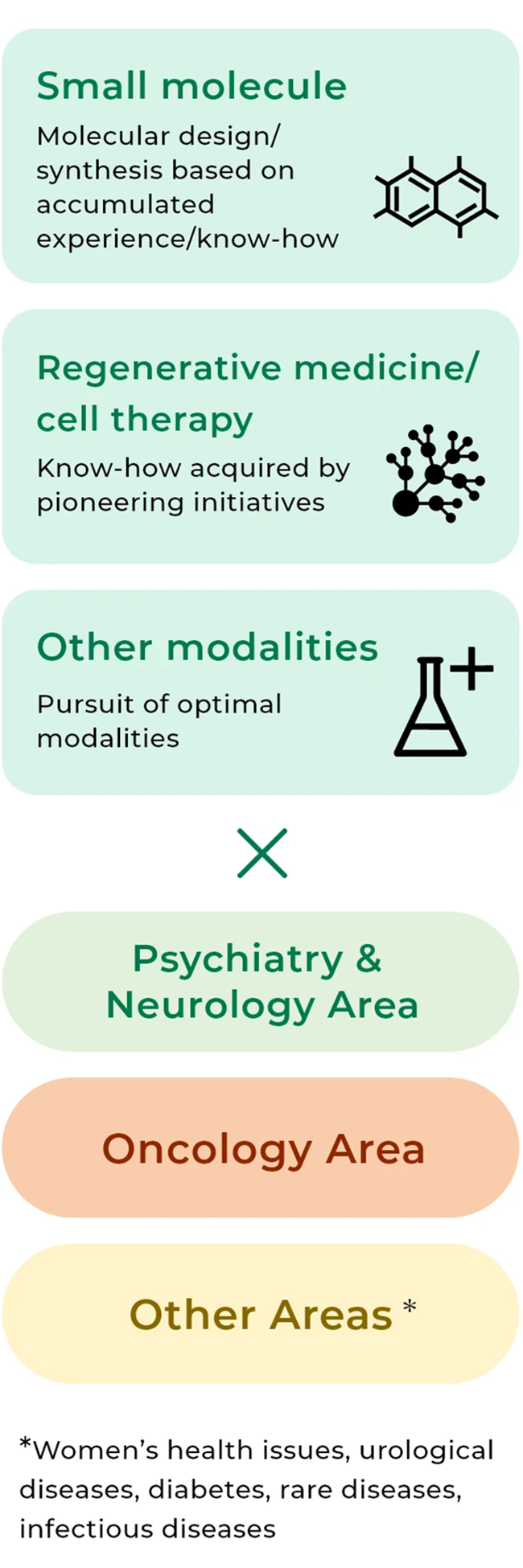
Our approach to the two priority disease areas and other areas
The Group aims to establish itself as a “Global Specialized Player” by 2033. With a focus on the Psychiatry & Neurology and Oncology disease areas, where unmet medical needs are high, we will make the utmost of the experience and knowledge that we have acquired so far to continue making active efforts for research and development of pharmaceuticals, regenerative medicine/cell therapy, non-pharmaceutical products, and otherwise. In other areas, too, we will tap into assets at hand and make steady R&D efforts to deliver solid value to patients. With regard to drug discovery research, we will forge ahead with our translational research and biomarker research and develop modality technologies to further strengthen our highly unique foundation for drug discovery. In addition, we will propel data-driven drug discovery to continuously create candidate compounds that focus on the essence of clinical conditions.
Psychiatry and Neurology Area
We are promoting competitive drug discovery research based on our proprietary drug discovery platforms established by continuously incorporating cutting-edge technologies. For psychiatric disorders, including schizophrenia, depression, and psychiatric symptoms related to neurological disorders, we aim to create innovative therapeutic agents that meet unmet medical needs through drug discovery based on neural circuit pathology, whereas for neurological disorders, including dementia, Parkinson's disease, and rare diseases, we seek to develop drugs for radical treatments of neurodegenerative diseases and other indications through drug discovery based on molecular pathological mechanisms. Every effort is being made to raise the success rate of research and development by applying the wealth of knowledge gained from clinical data of our products and development candidates, to translational research and by selecting appropriate drug discovery targets and biomarkers through the use of big data, such as genome information, brain waves, and imaging data. In FY2017, we introduced a new Research Project System, under which researchers who have devised project themes take the lead in their projects up to the initial clinical development stage as a general rule.
In the development stages, the Company is working closely with its U.S. subsidiaries under the global clinical development framework to expedite the receipt of approvals from regulatory authorities by efficiently promoting clinical development according to strategic development planning.
We are making steady efforts to prepare for the application for approval of allo iPS cell-derived dopaminergic neural progenitor cells for Parkinson’s disease in Japan and for the commencement of its clinical study in the U.S. by the end of FY2023. For allo iPS cell-derived retinal pigment epithelial cells, we will promote a program through a sponsor-initiated clinical study for retinal pigment epithelium tear that commenced in Japan in FY2023 in order to determine its therapeutic effects early.
For VR contents under development for social anxiety disorder (product code: BVR-100), we will work with our alliance partner to prepare for the commencement of its clinical study in the U.S. Meanwhile, for the wearable EEG meter, which is under development as a portable EEG meter for depression, we will make steady efforts to have it certified as a medical device in Japan.
Oncology Area
We have created multiple distinctive development pipelines as we gained a diverse array of knowledge to fortify drug discovery through our research and development efforts thus far. We leverage these unique pipelines to continue focusing on research and development of drugs in the Oncology area, where unmet medical needs are high.
For drug discovery, we aim to create innovative new drugs as we enhance our competitive edge by exploring new modalities with our proprietary technologies and conducting joint research with universities and research institutions.
In the development stage, we seek to improve the success rate for several products in our development pipeline that are under initial clinical evaluations by, for example, carefully assessing data in short-term, small-scale studies to identify cancer types optimally treated by them and determine the value of such products. In particular, we are concentrating our resources on TP-3654 and DSP-5336, aiming for obtaining an early approval.
Other Areas
We tap into assets at hand and make steady R&D efforts to deliver solid value to patients.
・For GEMTESA® (therapeutic agent for overactive bladder), we steadily proceed with application for approval of its additional indication for the treatment of overactive bladder in men with benign prostatic hyperplasia (BPH) in the U.S. and application for approval for overactive bladder in Europe, China, and Taiwan.
・To contribute to global health and precautions against future pandemics, we are engaged in research and development for the treatment of antimicrobial resistance (AMR), malaria vaccines, and a universal influenza vaccine.