Endeavors for Clinical Trials
Attitude of "challenge and creation"
To serve patients by delivering better new drugs as early as possible, we are promoting endeavors for early drug approval by preparing the systems and standards required for global clinical development and international joint testing.
On the other hand, it is a fact that we encounter many trials in the process of pharmaceutical product development. Even in these cases, we will continue to endeavor to achieve our mission by maintaining an attitude of "challenge and creation" so that we can be an "internationally competitive R&D-oriented pharmaceutical company."
Development organization aiming at higher safety
In clinical testing, we endeavor for the "promotion of development activities based on common sense and conscience" while always keeping in mind the risk of "unpredictable side effects." We secure the safety of cases by collecting, reporting and evaluating any kind of information, including that necessary for judging whether each case can cooperate with the trials, throughout the trial.
We have also arranged an in-house system for identifying the safety profiles of tested drugs promptly in the development stage in order to ensure higher safety in therapeutic trials.
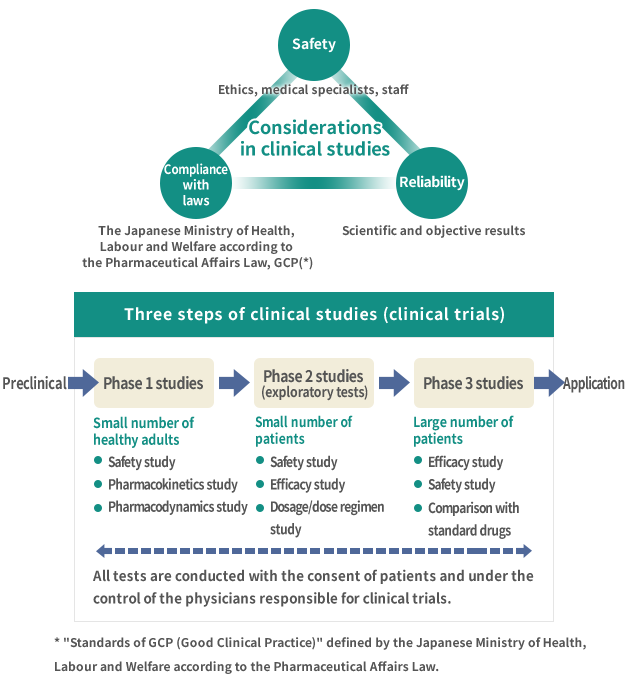
Image of Clinical Studies
What are clinical studies
A variety of drugs is used to cure a variety of diseases. These drugs are already in use after they were approved as "pharmaceutical products" by the national government (Ministry of Health, Labour and Welfare in the case of Japan). To let a substance that has newly been developed as a drug candidate, we conduct testing to check the efficacy (effectiveness) and safety (or side effects etc.) with cooperation of healthy people as well as patients of the target disease. The tests conducted under cooperation from the healthy people and patients are referred to as "clinical studies" and the clinical studies conducted in the process of obtaining approval of a drug candidate from the national government (Ministry of Health, Labour and Welfare) are referred to as "(government-level) clinical trials."
The (government-level) clinical trials are conducted in the following steps based on the Japanese Pharmaceuticals, Medical devices and Other Therapeutic Products Act and the "Standards on GCP" defined by the Ministry of Health, Labour and Welfare according to the law.
- ①The pharmaceutical enterprise and experts developing a drug candidate develop the "pharmaceutical approval trial program" upon discussion. This program is developed to evaluate the drug candidate optimally by taking the human right and safety in consideration.
- ②Upon receipt of the "pharmaceutical approval trial program" from the pharmaceutical enterprise, the Ministry investigates if the program takes the human right and safety in consideration.
- ③The hospital requested to perform the pharmaceutical approval trial by the pharmaceutical enterprise organizes an independent committee ("pharmaceutical approval trial committee") to check if the "pharmaceutical approval trial program" is scientifically appropriate and if it takes the human right and safety of the test participants (patients, etc.) in full consideration.
- ④The hospital in charge of the pharmaceutical approval trial should be equipped with sufficient facilities as well as the staffers including specialized physicians, pharmaceutical chemists, nurses and test coordinator.
- ⑤The physician in charge of pharmaceutical approval trial gives the participants the explanations of the test about the purpose, methodology, expected effects and possible side effects before starting the test. After it, the physician obtains documented "informed concept" from each patient participating the test from his or her own will.
- ⑥The privacy of people participating in the pharmaceutical approval trial is protected by law.
- ⑦If a side effect is observed with a person participating the pharmaceutical approval trial, necessary therapy and optimum treatment should be applied.
- ⑧The results obtained by the pharmaceutical approval trial (effects, side effects, etc.) are compiled by the pharmaceutical enterprise for use as materials so that the drug candidate obtain the governmental approval as a "pharmaceutical product."
What is "informed consent"
"Informed consent" refers to the action of a patient taken before receiving therapy or examination (including routine examinations) to "agree (consent) to receive the therapy from the patient's own will upon receiving sufficient explanation on the disease and its treatment policy by the physician, etc. and sufficient understanding of the details of the explanation by the patient oneself."
Informed consent in therapy
Since this stage is an intermediate stage for confirming the efficacy (benefit) and safety (side effects) of a drug candidate substance, it is a critical procedure that is indispensable for respecting and protecting the human right and safety of participants. Therefore, the "informed consent" in therapy should be performed in the following steps.
- ①The physician in charge of the pharmaceutical approval trial asks a patient meeting the prescription in the "pharmaceutical approval trial program" to cooperate with the test.
- ②When asking cooperation for the pharmaceutical approval trial, the physician hands the "explanation document" describing the purpose, methodology, expected effects and possible side effects to the patient and explain about its contents in details.
- ③Each patient brings the "explanation document" home to consult the family, etc., and determined whether or not the patient will participate in the therapy based on full understanding on it and from the patient's own will.
- ④If the patient decides to participate in the program, the patient signs the "document of consent" by oneself.