Product Development Research
To ensure that promising candidate compounds reach patients in the form of superior pharmaceuticals, we are committed to improving our product development technologies.
For continuously developing excellent pharmaceuticals and contributing to society, not only improvement in drug discovery ability or technology enhancement of clinical development but also enhancement of product development technology are indispensable.
We strive to develop pharmaceuticals that make the world a better place. Product development technology plays a key role in achieving this goal, right alongside drug discovery and clinical development.
The Company actively conducts product development research, including into new modalities such as bioprocess technology and new drug delivery systems, with the goal of making promising drug candidate compounds accessible to patients.
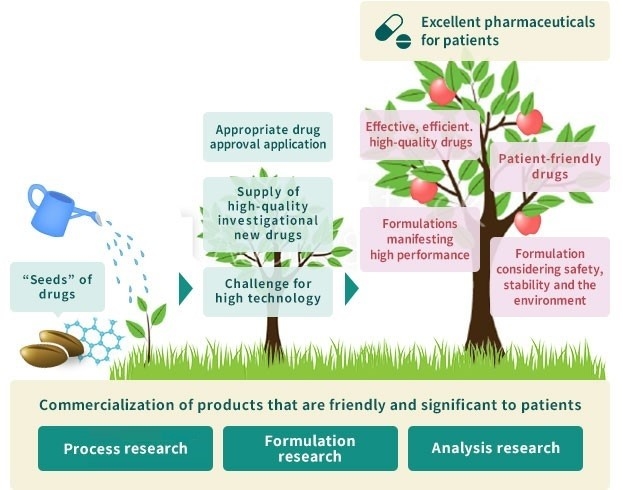
Product Development Research Process
Our product development research covers the whole lifecycle of a medicine, from the initial stages of drug discovery to post-marketing surveillance.

- Manufacturing/Supply/quality management of
investigational new drugs - New drug application
- Physical properties research
- Quality and stability evaluation research
- Basic technology research
- Formulation development research
- PLCM
- Route scouting
- Industrialization research
- Manufacturing process design
Bioprocess technology supports new modalities
The term "modality", which is frequently heard in the pharmaceutical industry, means a type of treatment (drug discovery technology and method). Conventional drug modalities were mainly small molecule drugs and antibody drugs. The Company focuses on cell therapy as a new modality, and is also working on functionalized proteins. Drawing upon the know-how in bioprocess technology it has acquired since the introduction of SUMIFERON®, the Company is promoting drug discovery research of new modalities based on the efficient manufacture of biopharmaceuticals. Furthermore, in the endeavor to discover new drug modalities using cutting-edge manufacturing technologies, the Company is pursuing the development of technology for protein functionalization through open innovation.
Drug discovery using new drug delivery systems (DDSs)
Discovery of new medicines is becoming increasingly difficult, but it is possible to reduce side effects and improve efficacy of an existing drug by changing its delivery system. This is called formulation discovery. Formulation technologies employed in formulation discovery can be conventional or new and innovative. To promote formulation discovery using innovative technologies, the Company is conducting joint research and open innovation projects with biotech companies and tertiary institutions. One example is DDS for nucleic acid drugs. Nucleic acid drugs such as antisense and siRNA are regarded as promising candidates for next-generation agents thanks to their excellent specificity and efficacy. However, innovation is required because, with the exception of the liver and certain other organs, at the present time, there are no DDSs capable of delivering drugs to specific target cells. In joint research into DDS formulation technology, the Company is conducting highly advanced research into the practical applications of nucleic acid drug delivery systems targeting patients for whom no effective treatment is currently available.
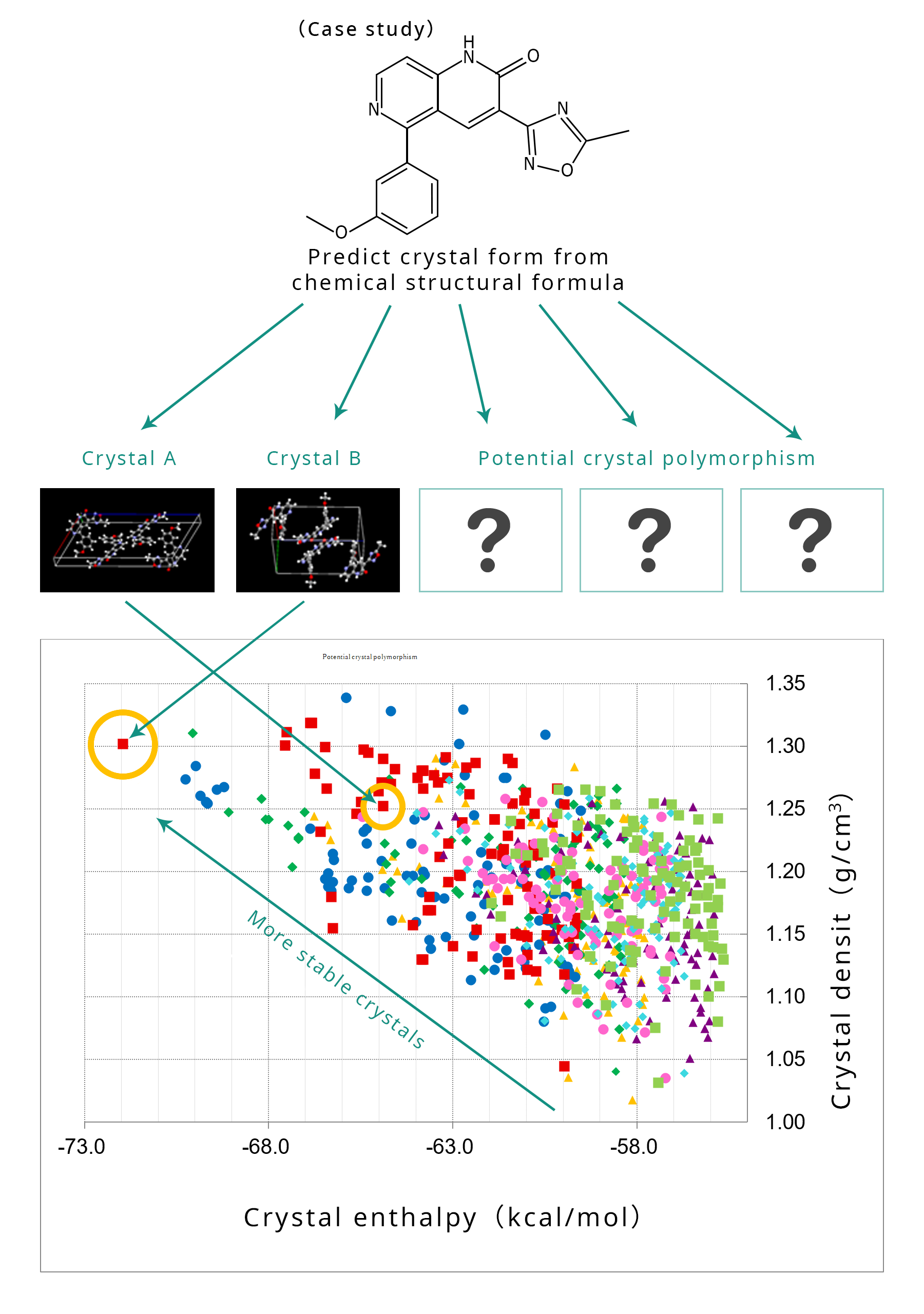
Prediction of quality using computer simulation
The Company is bringing computer simulation to formulation development. One example is crystal form choice. The crystal form of snow depends on temperature and moisture, and likewise, the crystal form of a drug substance varies depending on the manufacturing conditions. The optimum form is traditionally selected by "wet" screening experiments, but the Company is also researching computer simulation methods to evaluate whether the selected form is appropriate as a pharmaceutical. An additional example is predicting long term stability ("shelf life") of a drug by simulation. These simulations are based on short term stability data from the lab, but they allow us to ensure long term quality.
Please click here for examples of computersimulation.
Development of new formulation technologies to support maximization of product value
The Company is involved in the development of new formulation technologies to reach as many patients as possible. One of them is OPUSGRAN® technology, a unique formulation technology for solid oral preparations which allows the formulation of high-loading and high-sphericity granules of micrometer size of any drug regardless of its characteristics. With this technology, the Company is tackling the development of patient-friendly, high value-added formulations. It enables less frequent dosing, as it has excellent properties for sustained release. It also makes possible bitter taste masking and smaller tablets. Moreover, the Company has developed a unique nano-particle technology that can be applied to both oral and injectable preparations. In addition to these unique new technologies, various existing fundamental technologies are employed to develop new drugs and new high value-added drug forms, in cooperation with other companies, if necessary ("drug form deployment").
One successful example of drug form deployment is LONASEN® Tape, which was launched in September 2019. It is the world's first transdermal patch formulation indicated for the treatment of schizophrenia. Since the launch of LONASEN® Tablet and Powder in April 2008, the Company has been seeking to make LONASEN® more and more accessible to patients with a wider variety of needs. Formulation design to stabilize the blood concentration of the active ingredient blonanserin was conducted in cooperation with another company and resulted in a new treatment option which is responsive to the needs of patients.
Please click here for further information regarding OPUSGRAN® technology.
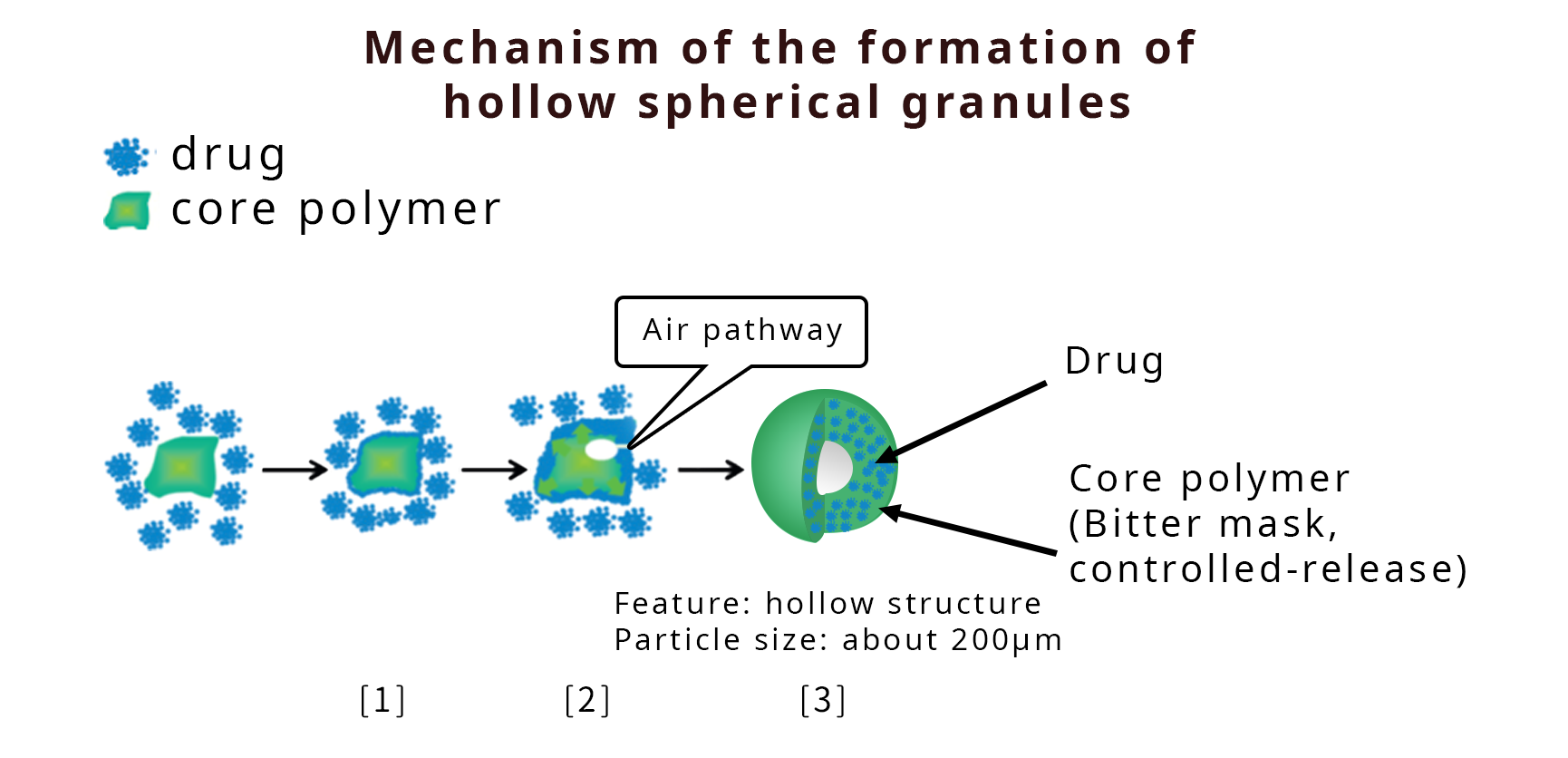
- [1] The granulation starts by wetting the polymer with sprayed solvent. Drug particles then stick to the polymer particle surface. Subsequently, a drug layer forms around the core polymer particle.
- [2] At some point, a crack forms in the adhered drug layer, and air flows into the granules.
- [3] The air makes a hollow, while the structure is kept without being crushed. The hollow expands as the granulation proceeds. Finally, the expansion of the hollow is completed, and the crack is closed by the compression force in the granulation. (Reference)Mechanism of the formation of hollow spherical granules using a high shear granulator. Eur. J. Pharm. Sci. 117,371-378 (2018)