Material Issues
In order to realize both contribution to sustainable society and its own sustained growth, the Sumitomo Pharma Group will create value that only it can deliver by utilizing the Group’s capital (strengths) in response to the diverse and changing social expectations and demands. To this end, in March 2023 we identified the key issues to be addressed as "Material Issues."
Material Issues Map
Process for Identifying Material Issues
STEP 1: Create a list of social issues/needs and the Sumitomo Pharma Group's capital (strengths)
We have made a list of social issues/needs by referring to such
things as the SDGs, Global Risk Reports, various frameworks (GRI
Standards, SASB Standards, ISO26000, The Ten Principles of the UN
Global Compact, etc.), and ESG survey evaluation items (DJSI, FTSE,
and MSCI).
As for the Sumitomo Pharma Group’s
capital (strengths), we made a list based on internal interviews and
by also taking into account information provided by institutional
investors.
STEP 2: Assessment of issues
Among the social issues/needs listed, we have extracted those issues
related to the Sumitomo Pharma Group's capital (strengths). Then we
have identified issues that are prioritized as Material Issues by
setting the following two axes for these issues: “Expectations
from society” and “Impact on corporate value
enhancement,” and we have assessed each of them on a
three-point scale.*
We conducted the assessment of issues by
also referring to advice from external consultants on
sustainability.
- *In terms of “Expectations from society,” we assess these issues based on the factors of “the level of public interest in the issue” and “the level of expectations towards pharmaceutical companies in addressing the issue,” and in terms of “Impact on corporate value enhancement,” it is based on the factors of “consistency with the Mission” and “the impact on corporate value through addressing the issue.”
STEP 3: Finalization of Material Issues
We finalize Material Issues after deliberations at multiple meetings of Management Committee and approval by the Board of Directors.
Targets and KPIs for Material Issues
To achieve both a sustainable society and our own long-term
corporate growth, we have identified our Material Issues and set
specific targets and KPIs for each. We regularly monitor progress
through KPIs and other indicators, make necessary adjustments, and
apply the PDCA (Plan-Do-Check-Act) cycle to ensure steady
advancement.
The Management Committee reviews and discusses
the progress and potential revisions of targets and KPIs for our
Material Issues at least once a year. The Board of Directors
deliberates and approves any revisions to the Material Issues.
We also regularly report progress to the Board, ensuring that
our initiatives based on the Material Issues are effectively
implemented under the Board's supervision.
In light of the current business environment and our fundamental structural reforms, we have recently revised certain targets and KPIs.
Historical Changes of Material Issues
-
Confirmation of Fiscal 2023 Progress (July 2024)
-
Identifying Material Issues and Confirmation of Fiscal 2022 Progress (July 2023)
The targets and KPIs of Material Issues and the status at the time of Material Issues identification (March 2023) (Integrated Report 2023, P91-94)
-
Confirmation of Fiscal 2021 Progress and Partial Revisions of KPIs (July 2022)
Materiality targets, KPIs and fiscal 2021 progress (Integrated Report 2022, P35-38)
-
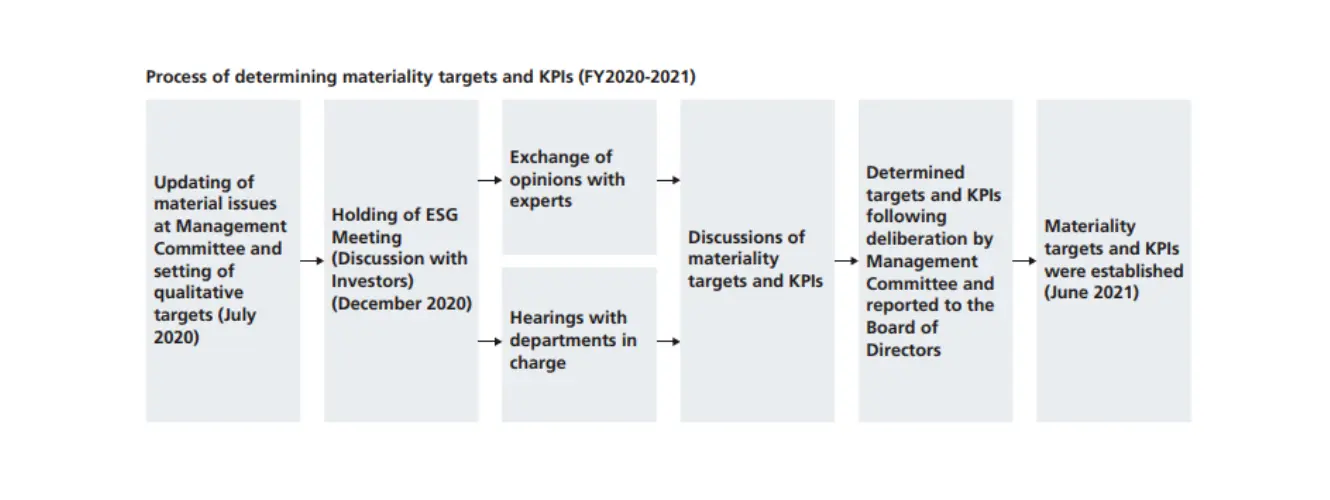
Establishing Materiality Targets and KPIs (June 2021)
We verified each of the material issues based on the opinions we obtained from dialogue with our stakeholders. As a result, we didn't change any of the material issues, and we established targets and KPIs for each of the material issues. In establishing such targets and KPIs, we ensured that they are linked to our business plans and management issues and made them indicators that enable us to evaluate and analyze the progress of each material issue. By sharing our initiatives with the public, including non-financial information, we further promoted dialogue with our stakeholders.
-
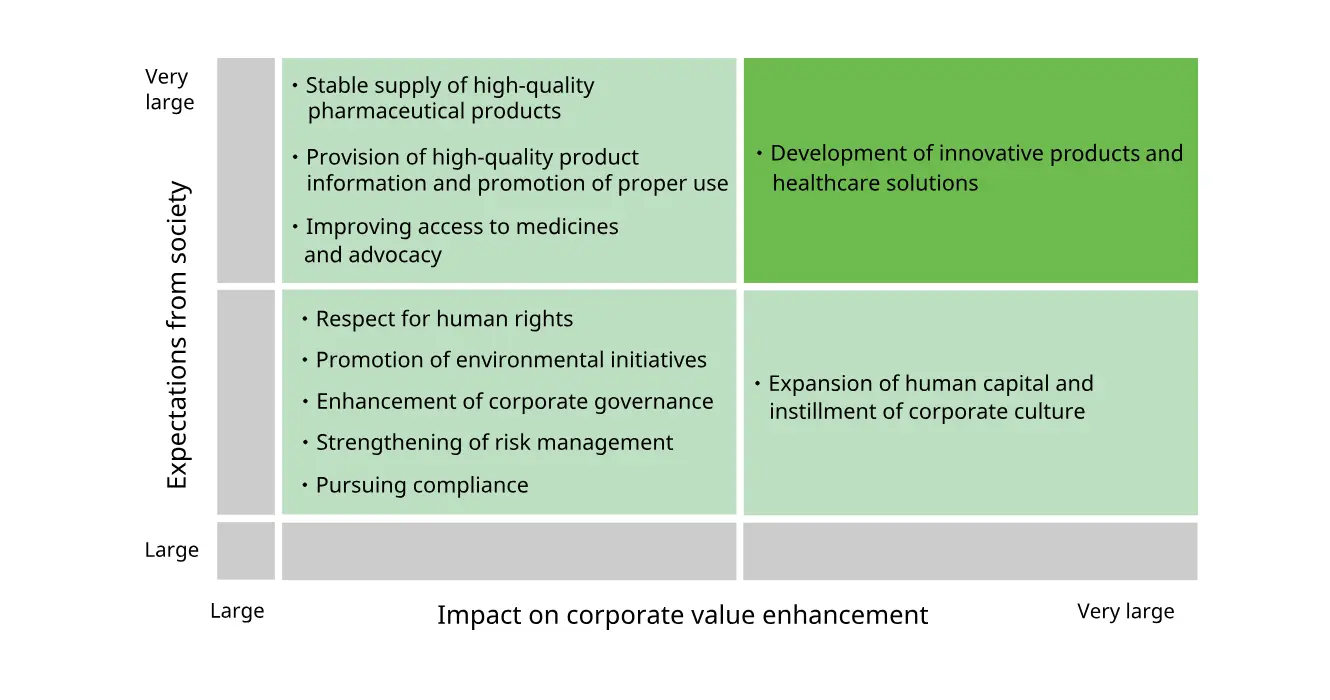
Changes to Materiality (July 2020)
Changes to Some Material Issues
- ①There are no changes in the area of "Material issues linked to value creation." "Development of innovative products and healthcare solutions" referred to psychiatry & neurology, oncology, regenerative medicine/cell therapy, infectious diseases, and frontier business, but we have deleted the reference to specific areas given that we will also focus on other areas in addition to these five.
- ②In the area of "Material issues that form the foundation for business continuity," given rising global concern in human rights, we have made an adjustment to further address respect for human rights through all of our business activities, and divided "CSR procurement (respecting human rights)" into "Respecting human rights" and "CSR procurement."
For more information about our initiatives on human rights. please see"Human Rights."
Establishing Materiality Qualitative Targets
In striving to achieve sustained growth and enhance corporate value, we have established qualitative targets for each material issue after a series of deliberations at the Management Committee. Because the present targets are a mix of Group-wide targets and targets in Japan, we have decided to continue revising them going forward to create more appropriate targets based on our business strategy.
-
Changes to Materiality (August 2019)
Based on stakeholder dialogue, we divided our materiality into the two categories below.
- ①Materiality linked to value creation, which is highly unique and vital for the sustained growth of Sumitomo Pharma
- ②Materiality that forms the foundation for business continuity, which is essential for the continuation of our business activities
Below are the reasons for the selection of the main material issues and how they are linked with value creation.
Development of innovative products and healthcare solutions, Contributing to the development of science
The development of innovative products and healthcare solutions to address unmet medical needs is our business and the source of value creation. We can also contribute to the development of science through drug discovery and research, product development, and clinical development.
Training and development of employees, Work Style Innovation, Diversity & Inclusion
A business is composed of its employees. Upgrading the capabilities of employees, improving labor productivity, and effectively utilizing human resources is essential for our sustained growth.
Contribution to global health, Initiatives to improve access to medicines
Sumitomo Pharma engages in drug discovery and research in the areas of malaria and antimicrobial resistant (AMR) infectious diseases. We also strive to improve access to our products for patients who need them, including responding to requests for development of unapproved and off-label drugs in Japan and public education aimed at improving medicine-related literacy. Such efforts contribute to global health in addition to expanding our business.
-
Initial Materiality Assessment Procedures (July 2018)
STEP 1: Compiling material issues
By referring to information* such as international guidelines and questionnaire factors evaluated by SRI assessment organizations,* we reviewed global societal issues and trends in ESG expectations to compile material issues which we should consider.
- *The United Nation's Sustainable Development Goals (SDGs), GRI Standards, ISO26000, Dow Jones Sustainability Indices, FTSE4Good, MSCI, etc.
STEP 2: Narrowing down material issues
We narrowed down the material issues based on the expectations obtained through dialogues with our stakeholders.
STEP 3: Prioritizing material issues
The material issues were classified and prioritized in terms of their "Societal Significance (based on the expectations of various stakeholders and social demand)" and "Importance to Sumitomo Pharma's Business."
STEP 4: Deliberating and determining material issues
We conducted materiality assessment through deliberation at the Management Committee to determine the material issues and shared them with all of the Directors in the Executive Committee.